Infographic
Italian perspectives on the safe handling of hazardous drugs and closed systems
This infographic and key paper summary explains how safe handling of hazardous drugs is approached in Italy
View and download
Infographic
EU strengthens protection of healthcare workers from hazardous substances
This infographic explains the changes to the Carcinogens, Mutagens and Reprotoxic Substances Directive, and how this will affect healthcare workers.
View and download
Editorial
Guidance for the management of hazardous medicinal products at work
Read our latest editorial summarising the key guidance applicable to healthcare professionals from the European Comissions guidance.
Read article
Infographic Content
CSTDs vs Closed Systems
Check out our infographic illustrating the differences between closed systems and CSTDs.
View and download
Editorial
Clearing up the confusion - closed systems versus CSTDs
This editorial explores the similarities and differences between closed systems and closed system drug transfer devices, explaining what they are, and why they’re used for safe handling of hazardous drugs.
Read article
Interactive Guide to the Guidelines
Contamination along the Care Continuum
This interactive pathway maps the key guidelines onto the various points of potential hazardous drug contamination in the hospital.
Explore the guidelines
Featured Content
Check out our latest videos
Watch Sara Arenas Lopez and Christian Reiss discuss the role of the pharmacist and waste management of hazardous drugs.
Watch now
About the site
Safe Handling of Hazardous Drugs
Discover helpful tips and resources on the safe handling of hazardous drugs. Check back regularly for new and updated resources including learning modules, infographics, factsheets, and expert videos.
Discover the resourcesFrontpage
Why use this platform

Topics tailored to your needs
Key questions modules are written by experienced pharmacists and cover important subjects to address the specific needs of a hospital pharmacist

Learn anytime, Anywhere
Hospital pharmacies are fast-paced environments; with these self-study modules, you can study when it’s convenient for you

New modules regularly added
Tell us what you want to learn.
We continuously add new modules, so you can update your knowledge in the areas that matter most to you

Fulfil CPD requirements
Earn points towards your annual CPD requirements by completing these self-study modules
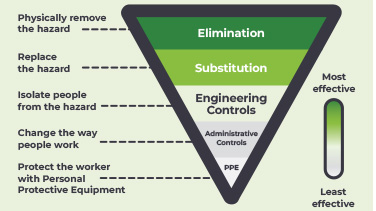
Engineering controls
Safe preparation and administration of hazardous drugs >
CSTDs vs Closed Systems
Engineering controls such as closed systems and CSTDs are essential for safe handling of hazardous drugs. This infographic illustrates the differences between closed systems and CSTDs.
CSTDs vs Closed Systems
Clearing up the confusion - closed systems versus CSTDs
Editorial article
This short editorial summarises the definitions of closed systems versus closed system drug transfer devices.
Clearing up the confusion - closed systems versus CSTDs
Reducing the risk of hazardous drug exposure to health care workers
Editorial article
One of the biggest concerns in the administration of hazardous drugs is the risk to those who come into contact with these drugs: patients, nurses and other health care professionals who administer the drug. Read this month’s editorial to explore ways of reducing these risks
Reducing the risk of hazardous drug exposure to health care workers
Surface contamination
Monitoring of hazardous drugs >
Environmental Wipe Sampling – Personnel and Practices
Editorial article
This editorial explains who is responsible for monitoring surface contamination of hazardous drugs, and why it is so important
Environmental Wipe Sampling – Personnel and Practices
Precautions, perspectives, and practicalities: the reality of safe handling of hazardous drugs in hospitals
Editorial article
This short article summarises the main barriers to safe handling of hazardous drugs from the perspectives of nurses
Precautions, perspectives, and practicalities: the reality of safe handling of hazardous drugs in hospitals
Monitoring contamination of hazardous drug compounding surfaces at hospital pharmacy departments
Key Paper Evaluation
This 2-page evaluation summarises key findings from a review of surface contamination monitoring in hospital pharmacy departments conducted by the Spanish Society of Hospital Pharmacists (SEFH).
Monitoring contamination of hazardous drug compounding surfaces at hospital pharmacy departments
Guidelines and directives
Evidence base for safe handling of hazardous drugs >
Italian perspectives on the safe handling of hazardous drugs and closed systems
Infographic
This infographic and key paper summary explains how safe handling of hazardous drugs is approached in Italy.
Italian perspectives on the safe handling of hazardous drugs and closed systems
Carcinogens, Mutagens and Reprotoxic Substances Directive
Authors: Susanne Cruikshank & José Manuel Martínez Sesmero
The Carcinogens, Mutagens and Reprotoxic Substances Directive (CMRD) is entering into law in April 2024. It contains guidance for employers on how to keep healthcare staff safe at work when handling hazardous medicinal products. Watch this conversation between Susanne Cruikshank and José Manuel Martínez Sesmero to understand more about the importance of the CMRD.
Carcinogens, Mutagens and Reprotoxic Substances Directive
EU strengthens protection of healthcare workers from hazardous substances
Infographic
This infographic explains the changes to the Carcinogens, Mutagens and Reprotoxic Substances Directive, and how this will affect healthcare workers.